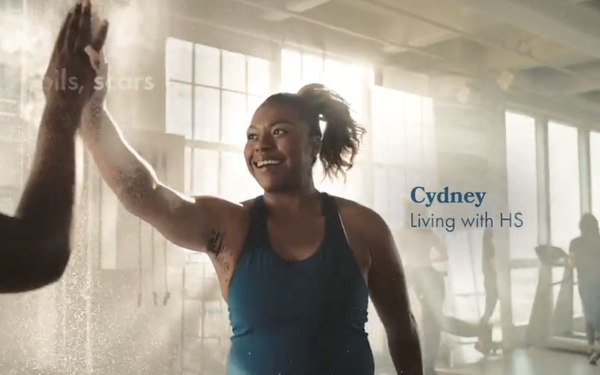
Hidradenitis suppurativa (HS), a not-so-rare skin disease, is coming
out of the closet, so to speak.
Affecting an estimated 3.3 million Americans, HS manifests itself through permanent scarring, caused by boil-like abscesses, that most often occurs in skin
creases like armpits, the groin area, and the buttocks. Other symptoms include intense pain, pus discharge and odors.
The symptom locations and associated embarrassment have helped lead
to long delays in patients getting diagnosed, Charity Williams, patient marketing director for pharma firm UCB, tells Pharma & Health Insider. There are also a lot of misdiagnoses before HS
is finally determined as the cause, she notes.
To educate both patients and dermatologists about the disease, UCB is running “Make HStory,” an unbranded educational campaign from
Havas Tonic that aims to help increase understanding of the disease and to empower patients and their doctors to have conversations about managing the condition.
advertisement
advertisement
The consumer part of the
campaign launched earlier this month, a year after UCB began targeting physicians. It includes a website; a :30
spot running on connected TV services like Hulu, Netflix and YouTube; and paid ads on Meta platforms. PHM is the media agency.
The main target audience is 25- to 50-year-olds, Williams
says, with “a wide range of diversity.” She notes that HS occurs more often in women and in people of color. Symptoms usually start at puberty and have their biggest impact on people in
their 20s and 30s. Symptoms tend to be less intense once patients reach their 50s and 60s, she adds.
UCB isn’t alone among pharma companies in efforts to generate HS awareness, with
similar unbranded awareness campaigns having been launched in the past few years by both Abbvie and Novartis.
The Novartis campaign, “Shine a Light on HS,” launched in 2022, a year before its psoriasis drug Cosentyx received FDA approval to also treat HS.
Abbvie’s campaign, “No BS About HS,” launched in 2021. The company currently has an HS drug
in Phase 3 clinical trials.
In UCB’s case, the drug awaiting FDA approval for HS is Bimzelx, approved for psoriasis late last year, and already the star of a “Get Yourself
Back” campaign that launched last month during ABC’s Oscar telecast. UCB expects approval of Bimzelx for HS by the end of
2024.
No drug was approved for HS by the FDA until 2021, when Abbvie’s blockbuster anti-inflammatory Humira, now available under different names from numerous companies, received that
designation. Here’s Humira first HS commercial.
Despite the biologics already on the market, HS is still largely treated
with topicals, antibiotics and “a high prevalence of home remedies that patients use from talking to family members and figuring things out on their own,” says Williams.
Now,
though, says UCB in its new spot showcasing actual HS patients, “It’s your time to make ‘HStory,” to swap feeling body conscious for a body con dress, and let the sparks fly,
to move away from the isolation, the worry of pain, boils and scars. It’s your moment to feel good in your skin again.”
Williams says that, rather than use lots of marketing
research to develop the campaign, “We brought in people living with HS” as co-creators. “We actually had them physically put hands on our campaign,” she explains. “I
think that’s what’s going to make this have such a huge impact….It wasn’t just something that we as marketers felt was the right message. It was really created by people that
need it the most.”